Assisting the failing heart with muscle power
A team of CMU researchers is developing a self-contained, non-blood-contracting ventricular assist device that uses muscle power to support the failing heart.
Heart disease is the number one cause of death in the United States. According to the Columbia University Department of Surgery, 5.8 million Americans have some level of heart failure, and about half of patients die within five years of diagnosis.
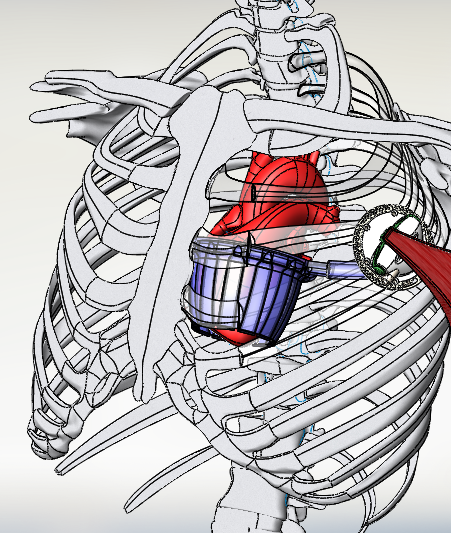
Source: Dennis Trumble
Internal muscle energy converter (MEC) and direct cardiac compression sleeve (DCCS)
“There are currently three types of treatments out there, but drugs and heart transplantations have their limitations,” said Jooli Han, a doctoral student in biomedical engineering at Carnegie Mellon University. “Many chronic heart failure patients use ventricular assist devices (VADs) to restore normal blood flow, but these have two main problems. One is the risk of infection from drivelines that pierce the skin, and the second is potential blood clotting due to the bloodstream contacting artificial surfaces inside the pump.”
Is there a way to help those patients without introducing infection risks or interfering with their blood? To turn that dream into reality, Han is working with Dennis Trumble, an associate research professor of biomedical engineering, to develop a self-contained, non-blood-contracting VAD for long-term use. Their project is supported by a grant from the National Institutes of Health.
This VAD helps the heart pump blood by using muscle power. It uses an internal muscle energy converter (MEC) to drive a direct cardiac compression sleeve (DCCS) attached outside the ventricles. The MEC is a hydraulic actuator designed to attach to the latissimus dorsi, a large muscle on the back. When the heart beats, a muscle stimulator causes the muscle to contract, which rotates the actuator arm of the MEC. Fluid is then ejected through the outlet port of the device and enters the sleeve, which squeezes on the heart and helps it do its job.
If we condition them in the right way, we can create a muscle that will contract repeatedly without fatigue like a marathon runner.
Dennis Trumble, Associate Research Professor, Biomedical Engineering
“We’ve learned to train the muscle electrically,” said Trumble. “Muscles are very plastic and able to adjust their anatomy and function over time in accordance with how they are used. So if we condition them in the right way, we can create a muscle that will contract repeatedly without fatigue like a marathon runner.”
Using ANSYS, an engineering simulation and three-dimensional (3D) design software package, Trumble’s team has tested and adjusted their designs of the VAD. The ANSYS Workbench Finite Element Analysis (FEA) tool allows engineers to analyze multiple design scenarios and customize solutions to solve structural problems.
After drawing a 3D model of their device, Trumble’s team imported the model into ANSYS and applied its material properties in the software. Then, they set functional boundary conditions by adding fixtures and different levels of pressure. From there, they could see how the model deformed as they changed the amount of pressure applied to the interior surfaces of the sleeve. They adjusted the model until they achieved the desired result and built the device to test whether it would reflect the simulation.
“We’ve been using ANSYS for years now,” said Han. “It’s a big part of our research, and it’s especially useful for soft robotics development like what I do. It’s important to simulate what would happen before I spend so much time and material to make it.”
Currently, Trumble’s team is in the process of developing its soft robotic cardiac sleeve device. Collaborating with a lab from Johns Hopkins University, they are using different types of soft material to 3D print the sleeves and test them. They aim to find the best material to maximize the output of muscle-assisted heart contractions.
Their ultimate goal is to customize these devices to meet the needs of individual patients; the one-size-fits-all generic compression of the heart might not help everyone as no two heart failure cases are exactly the same. To help all patients optimally, they plan to examine individuals’ hearts, conduct simulations to find the kind of compression that meets their needs, and build the device accordingly.
